A new federally-backed blood test promises instant disease detection for just $2, but raises urgent questions about data privacy and government overreach in American healthcare.
Story Highlights
- ASU scientists unveil NasRED, a $2 nanotech blood test that detects deadly diseases in 15 minutes.
- The portable device requires no lab infrastructure, making rapid diagnosis accessible nationwide.
- Supporters tout public health benefits, but critics warn of risks to privacy and centralized data control.
- No roadmap yet for regulatory approval, commercial deployment, or protection of individual rights.
Revolutionary Diagnostic Technology: Speed Meets Simplicity
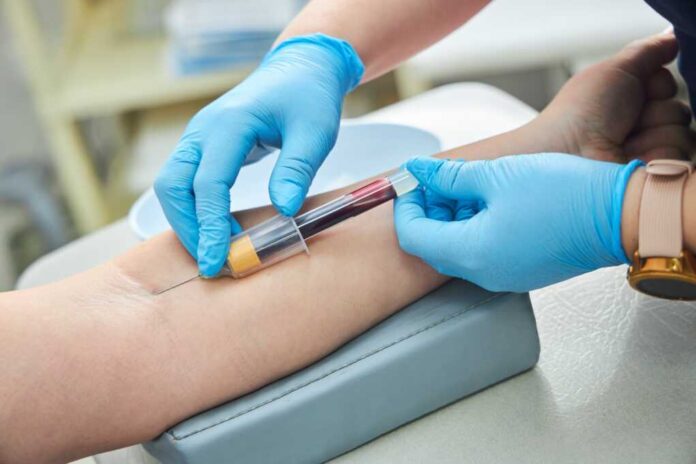
Arizona State University’s research team has developed NasRED, a game-changing blood test that delivers lab-quality results for diseases like COVID-19, Ebola, HIV, and Lyme disease with just a single drop and a 15-minute wait. The device, costing a mere $2 per test, is portable and requires no specialized training or laboratory equipment. This breakthrough aims to democratize access to high-quality medical diagnostics, especially for rural and underserved communities, and could transform how America responds to public health threats.
Watch a report:
The COVID-19 pandemic exposed glaring weaknesses in the nation’s diagnostic infrastructure, particularly in low-resource regions. Traditional PCR-based tests, though accurate, demand significant time and centralized facilities. By contrast, NasRED’s nanotechnology and biosensor design enable high-sensitivity detection on the spot, addressing a major barrier to effective outbreak control and patient care. The test’s affordability and ease of use position it as a potential equalizer for communities historically left behind by government and bureaucratic inefficiencies.
Who Holds the Power? Stakeholders, Motivations, and Government Reach
Arizona State University, led by Chao Wang and his multidisciplinary team, stands at the forefront of this innovation, driven by a mission to improve global health and advance scientific progress. While healthcare providers and public health agencies see value in rapid diagnostics for better patient outcomes and outbreak response, the device’s success depends on regulatory approval and future partnerships. With the Trump administration emphasizing state control and limited federal oversight, questions arise about who will manage data collection and privacy if NasRED sees national deployment.
Regulatory agencies like the FDA and potential industry partners will play pivotal roles in determining how and where NasRED is implemented. The Trump administration’s recent moves to return healthcare and education oversight to the states highlight both opportunities for local empowerment and risks of inconsistent standards. As with previous government-led health initiatives, the handling of personal medical information and deployment logistics could become flashpoints for debate, especially among conservatives wary of government tracking and federal data mandates.
A $2 gold nanotech test that detects deadly diseases in minutes https://t.co/NYjzUyiDHg
— Lifeboat Foundation (@LifeboatHQ) August 23, 2025
Current Developments and Unanswered Questions
NasRED has demonstrated remarkable accuracy in research settings, outperforming many rapid antigen tests currently in use. However, despite the media fanfare and peer-reviewed publication in ACS Nano, there is no public information yet on regulatory approval, commercial rollout, or large-scale production. Ongoing research aims to expand validation to additional diseases, but the lack of a clear deployment roadmap leaves Americans uncertain about when—or if—this technology will reach their communities without strings attached.
Another critical concern is the device’s data management. Will patient results be stored locally, or transmitted to centralized databases? Who will access this information, and how will individual privacy be protected in an era of increasing digital surveillance? Without strong constitutional safeguards, the risk of government overreach or unintended consequences remains real, especially as prior administrations have pushed for centralized health data systems in the name of public safety.
Sources:
ASU News, August 12, 2025: Fast, accurate, low-cost diagnostics: No lab required
ASU News, March 24, 2025: College of Health Solutions launches first-of-its-kind diagnostics industry partnership
Phoenix Bioscience Core: ASU National Diagnostics Summit 2025